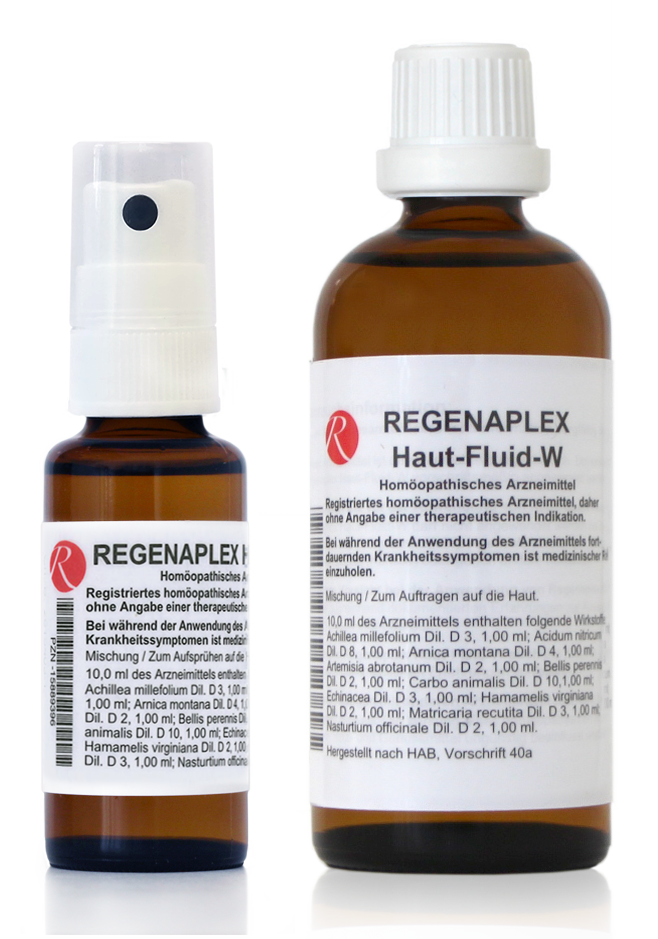
REGENAPLEX Haut-Fluid-W
10 ml of the medicinal product contains 1 ml each of:
Achillea millefolium D3 (HAB), Acidum nitricum D8 (HAB), Arnica montana D4 (HAB), Artemisia abrotanum D2 (HAB), Bellis perennis D2 (HAB), Carbo animalis D10 (HAB), Echinacea D3 (HAB), Hamamelis virginiana D2 (HAB), Matricaria recutita D3 (HAB), Nasturtium officinale D2 (HAB).
Instructions for use:
What do you need to know before using Regenaplex Haut-Fluid-W?
Regenaplex Haut-Fluid-W should not be used in case of known hypersensitivity to Achillea millefolium (yarrow), Arnica montana (arnica), Artemisia abrotanum (boar eye), Bellis perennis (daisy), Echinacea (sun hat), Matricaria recutita (chamomile) or other composite flowers.
Only use Regenaplex Haut-Fluid-W for children under the age of 12 after consulting the doctor, because there are no sufficiently documented experiences.
No interaction with other medicinal products are known.
The effect of a homeopathical remedy can be influenced negatively by life-style factors as stimulants and luxury food. If you take any other medication inform your healthcare professional accordingly.
How to apply Haut-Fluid-W?
Unless otherwise prescribed, apply Regenaplex Haut-Fluid-W 2-3 times daily to affected skin areas.
Reduce the frequency of use for once the symptons reduce.
Note: The product is for external use only. Contact with the eyes and mucous membranes, as well as open wounds should be avoided.
If your symptoms get worse or there is no improvement after a few days, be sure to call a doctor.
If you have any further questions on the use of this medicine, ask your doctor or pharmacist.
What side effects are possible?
In rare cases, hypersensitivity reactions may occur. Local allergic reactions (inflammation of the skin) have been reported. In these cases you should stop taking the medicine and see your doctor. Bei längerer Anwendung sollte auf UV-Bestrahlung sowie intensives Sonnenbaden verzichtet werden. Prolonged application UV irradiation as weill as intensive sunbathing should be avoided.
Note: When using homeopathic medicines, existing symptoms may temporarily worsen (initial worsening).
In this case you should stop taking the medicine and consult your doctor.
If you notice any side effects not listed in the leaflet, please tell your doctor or pharmacist.
You can also report side effects directly to the Federal Institute for Drugs and Medical Devices, Dept. Pharmacovigilance, Kurt-Georg-Kiesinger Allee 3, D-53175 Bonn, website: www.bfarm.de. By reporting side effects, you can help provide more information about the safety of this medicine.
Other warnings: Contains 51 vol.% Alcohol
